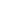Does Brass Rust? Corrosion, Oxidation, and Care
 Published:Nov 29,2024
Published:Nov 29,2024

Brass is a metal blend of zinc and copper with 60/40% or 70/30%, respectively. The composition varies depending on the application, which thus affects the corrosion properties. The corrosion phenomenon in brass is different from steel. like steel can be corroded when exposed to water but brass does not. This article will discuss in detail the corrosion resistance and function of protective layer of brass.
Will Brass Rust, or Is It a Timeless Material?
No, brass does not corrode or erode. Brass is a timeless material with no corrosion. It can last up to centuries if proper care is taken. Brass has quality to maintain its integrity in moisture and humidity. That is why it is suitable for kitchen and bathroom applications.

Rust vs. Corrosion
Rust is a specific term of corrosion for iron and alloys such as steel. When iron containing material reacts with oxygen, a chemical reaction occurs. This turns the metal blend reddish-brown in color and a flakey. This process in iron oxides is called rust.
While corrosion is a generic term used for any materials, such as metals and non-metals. In this process, the surface of material deteriorates when reacts with oxygen, hydrogen, electrical current or any dirt or bacteria. The resultant surface deterioration is called corrosion.
Does Brass Corrode?
Yes, the surface of brass can corrode. The corrosion occurs when the surface is exposed to moisture, acetates, ammonia, chlorides, and certain acids.
Does Brass Oxidize?
Yes, brass oxidizes. The surface of brass gets eroded, discolored, corroded, stained, and tarnished. This process is known as oxidation. A natural film called Patina is developed over the surface and it discolors the brass.
Is brass oxidation harmful?
Yes, sometimes it can be harmful. some of the brass components contain lead. When oxidation occurs, it releases metal to drinking water.
Is Brass Rust proof?
Yes, brass is rust-proof. It does not contain iron or chromium oxide like steel, which is important for the occurrence of rust. However, a Patina layer is developed over the surface which discolors the brass when used in outdoor applications.
Does Brass Change Color?
Yes, brass changes color with time due to corrosion. Corrosion in brass occurs by dezincification. This causes the brass to lose zinc and changes its color from yellow to pink. If it is severe, it weakens the brass and causes perforation.
Does Brass Turn Green?
Yes, brass turns green due to oxidation. A layer called Patina is developed that is a sign of aging. This is oxidation on the surface of brass and turns the brass green.
Why Brass Doesn’t Rust Like Iron?
Brass does not rust. It does not have iron in it or chromium oxide which can cause rust. But copper in brass can form blue or green patina when it starts losing zinc due to oxidation.
The Role of Copper and Zinc in Corrosion Resistance
The metal blend of Cu and Zn in brass provides different kinds of resistance. Zn provides higher hardness and mechanical resistance. while Cu gives corrosion resistance. Zn is more reactive and prone to corrosion than Cu. So, whenever oxidation occurs, it targets Zn. If zinc is more than 15% in metal blend, it can cause dezincification. This weakens the copper layer and corrosion starts immediately.

Factors Affecting Brass Corrosion
Brass corrosion occurs when the surface reacts with oxygen, water, chemical like ammonia, chloride, bacteria or dirt, or other environmental factors. Variation in temperature or high humidity immediately oxidizes the surface. If pollutants contain compounds like sulphur, severe corrosion can occur. Similarly, presence of salt, ammonia or any acidic solution can start corrosion in brass.
What Is Dezincification?
Dezincification occurs when brass starts losing zinc and the protective layer. It occurs by an electrochemical reaction in which zinc reacts with chemicals in water. This causes a weak spongy layer of Cu on the surface.
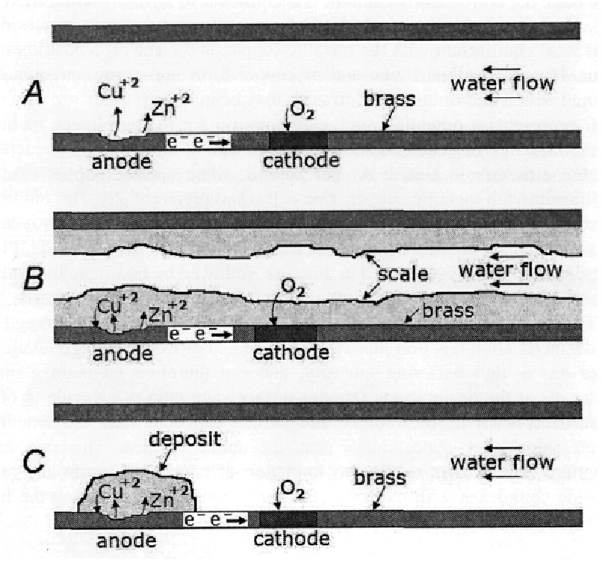
Dealloying Explained
Dealloying can be seen from the naked eye. It turns the surface reddish-brown which is different from its natural yellowish color. When dealloying occurs, the protective layer vanishes, and dull spots start occurring over the surface and block the passageways.
Impact on Structural Integrity
Dezincification can have different impact on the surface of brass depending on the corrosion conditions. If it is severed, it can cause actual breakage and leave a dull coppery appearance at the fractured surface.
How Dezincification Alters Appearance
Dezincification alters the color but does not change the dimensions. It normally appears as a white spot of zinc oxide on the surface. and sometime the mineral stains appear on the surface. Water leakage also occurs from the valve body.
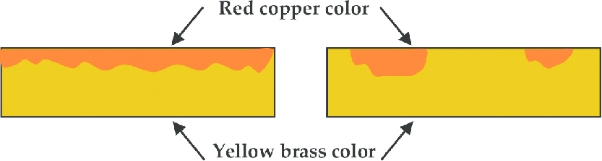
Examples of Brass Corrosion in Real Life
Brass is one of the commonly used alloys in our daily life. The common examples of brass corrosion are:
Household Fixtures and Fittings
Brass fixtures and fittings have high corrosion resistance. That is why it is the best choice for plumbing and household applications. However, fixtures can erode and change color with time. A patina layer forms over the surface and turns it green.
Marine Environments
Brass oxidizes immediately when exposed to saltwater. However, its structural integrity does not get affected much. Corrosion of brass is higher in natural seawater as compared to artificial seawater. The comparison is made with respect to pH, contact with oxygen and other marine environmental factors.
Industrial Applications
Brass is normally used when low friction and high corrosion resistance are required. The common applications are gears, bearings, locks, hinges, ammunition casings, hose coupling, zippers, electrical plugs, sockets and plumbing. The chance of corrosion is less but exposure to humidity can tarnish brass in these components.
How to Prevent Brass Corrosion
There are a few ways to protect brass from corrosion. Some of them are:
Coatings and Protective Finishes
Apply a thin protective layer of tin, Nickle, zinc, or polymer to the brass. This creates a barrier and protects it from corrosion. Chrome plating also protects the brass from humidity and chemical reaction. Wax coating protects it from handling and common environmental factors.
Regular Maintenance Practices
Maintain cleanliness by removing dust regularly over the brass surface. mild soap and water can clean the dust from surface .Thoroughly rinse and dry with soft cloth. Commercial polish must be avoided because it contains acids or ammonia. Use gloves when handling brass to prevent dezincification.
Avoiding Harsh Chemicals
The surface of brass must be protected from harsh chemicals, or excessive moisture. Some cleaning agents contain acids or chlorides that can trigger corrosion. Harsh abrasive must be avoided for cleaning like steel wool.
How to Restore Tarnished Brass
There are a few ways to restore tarnished brass. This includes commercial brass cleaner, mild soap and water, vinegar and salt and backing soda. Proper instruction must be followed and specified quantity of cleaning agent must be used. Prolong contact with cleaner or excessive quantity can further damage the brass. Use cleaner on a small, inconspicuous area first.
How do I Get Rust off of Brass?
Commercial rust remover can be used by following instructions. Other ways are using mild soap and water, vinegar and salt and backing sod on the corroded and eroded surface of brass.
How to Clean Brass Hardware
Brass hardware can be cleaned by mixing parts vinegar, salt and flour to make a paste. Then clean the brass hardware by gently rubbing with a soft brush. Finally, rinse and wipe with damp cloth.
How to clean Brass Jewelry
Brass jewelry can be cleaned by making a paste of vinegar, salt and flour. And then rub and clean the jewelry with the paste gently.

Conclusion
Understanding brass and its rust resistance is crucial for manufacturing and mechanical design. Knowing its composition, properties, and corrosion resistance aids in application decisions. Regular maintenance and protective coatings prevent tarnish and extend lifespan. Tuofa CNC machining services in China can enhance brass component fabrication and durability. Click here to get a free quote.
 Tel/WeChat:
Tel/WeChat:  Email:
Email: 
 Home
Home
 What are Uses of Aluminium? Everything You Should Know
What are Uses of Aluminium? Everything You Should Know